これ以降の情報は医療機関にお勤めの医療従事者を対象として提供しております。
一般の方に対する情報提供を目的としたものではありませんので予めご了承ください。
あなたは医療従事者ですか?
-
はい
- いいえ
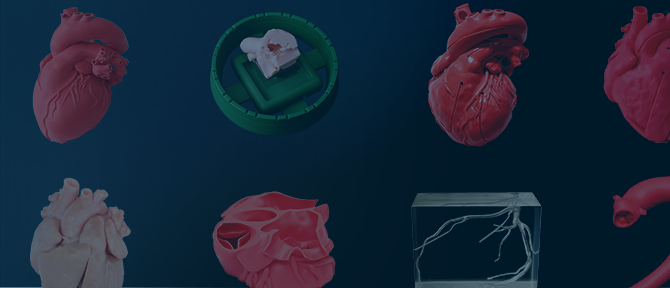
心疾患患者様の術前診断を
サポートする立体臓器模型
心疾患患者様一人ひとりのCTデータをもとに製作したCARDIO Simulatorは、
心臓の解剖学的構造を把握することができる医療機器です。
実物大の立体臓器模型のため、異常部位の場所や大きさ、周辺組織との位置関係などが明らかになり、
術前の診断を補完することが可能です。
- 製品名:術前支援モデル「CARDIO Simulator」
- 医療機器届出番号:26B2X10032000001
特長
-
 01
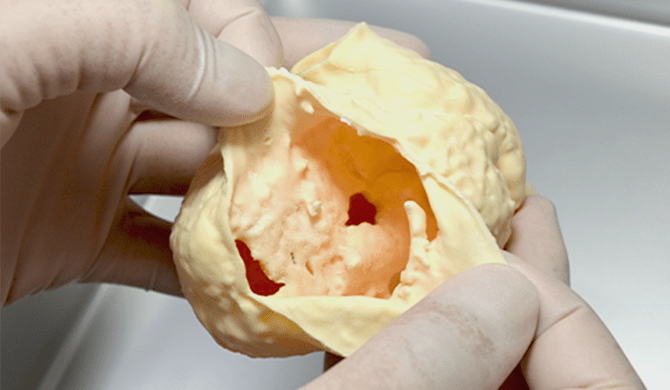
01個々の生体データより得られる
情報を立体的に表現 -
 02
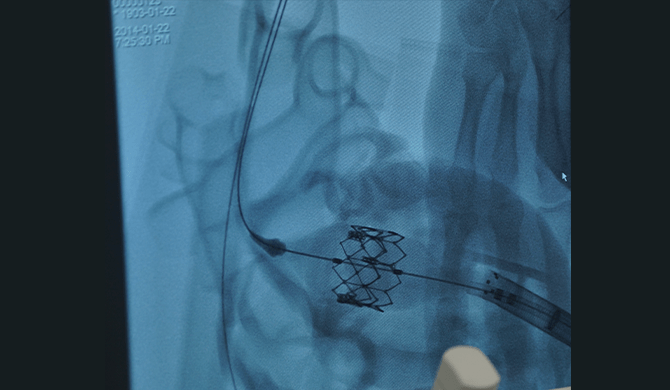
02各組織の形状や部位間の
距離などを実物大で製作 -
 03
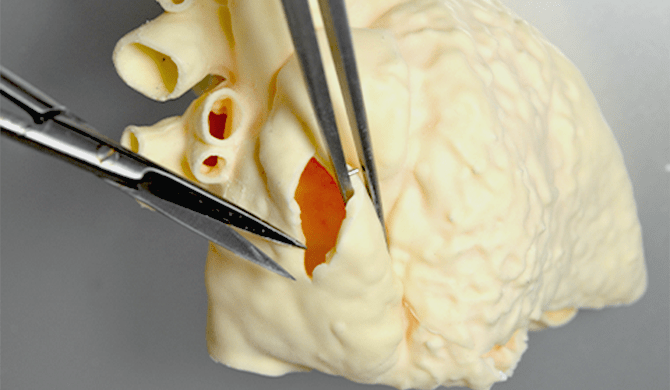
03様々な角度から
重要箇所を確認することにより
詳細な構造把握が可能 -
 04
04軟質樹脂や硬質樹脂などの
硬さに加え透明や有色など
様々な素材の選択が可能
使用目的
外科目的
- 心内リルーティングなどが必要な症例について、画像診断だけでは理解が不完全な場合に、「CARDIO Simulator」を活用したディスカッションを行ない、より理解を深めたい。
- 手術に臨むにあたり、画像診断だけでは十分な医療情報が得られていない可能性がある場合に「CARDIO Simulator」を用いて、より理解を深めたい。
- 画像診断で施術方針の選択が難しい症例について、「CARDIO Simulator」を活用したい。
- 心内修復術を行なうなど、術前術後に形態の変化が大きい症例に対しての評価に活用したい。
内科目的
- 画像診断だけでは難しい症例の診断に対して、「CARDIO Simulator」を活用したディスカッションを行ない、
より理解を深めたい。 - 画像診断ではカテーテルを用いた施術が可能であるか判断が難しい場合、「CARDIO Simulator」を活用して理解を深めたい。
- 画像診断だけでは診断が難しい場合、「CARDIO Simulator」を補助的に活用したい。
- 心内構造をより深く理解したい。
仕様
フルタイプ
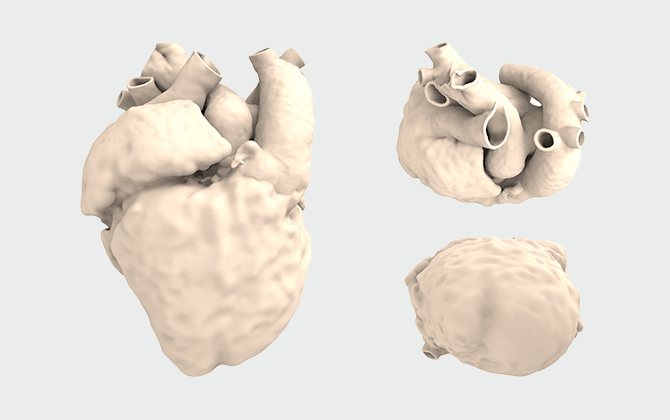
上
下
正面
| 目的・ 使用用途 |
対象症例の全体的な形態確認 |
|---|---|
| 範囲 | 血管/心房/心室 |
心房タイプ
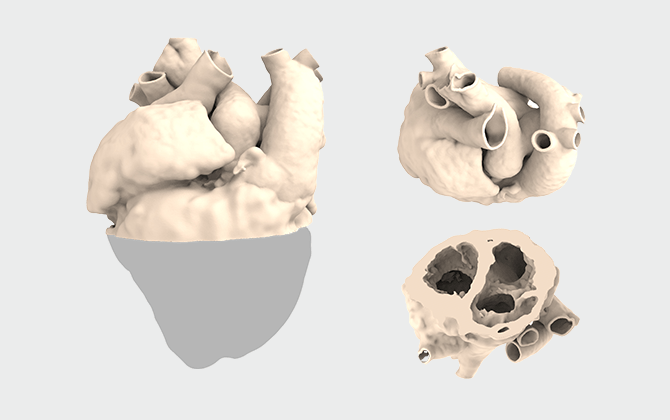
上
下
正面
| 目的・ 使用用途 |
対象症例の血管および弁周辺の形態確認 |
|---|---|
| 範囲 | 血管/心房/心室の一部 |
心室タイプ
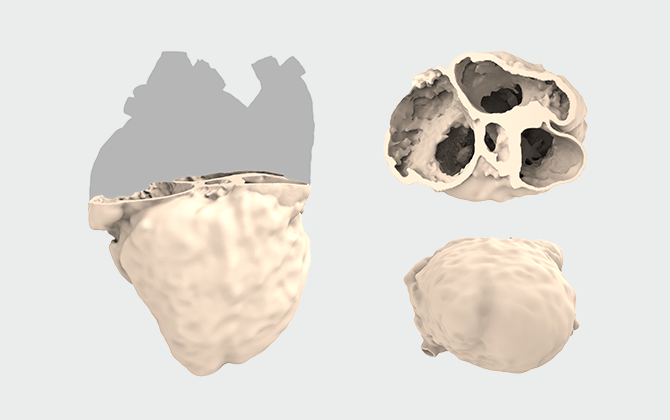
上
下
正面
| 目的・ 使用用途 |
対象症例の弁周辺および心房、心室の形態確認 |
|---|---|
| 範囲 | 心房の一部/心室 |
- 症例により、仕上がりは異なります。詳細はMSCTデータ(DICOM形式)を確認後、担当者よりご連絡します。
- 価格は、サイズによって異なります。サイズのおおよその目安は次の通りです。
- [S] 約4 x 4 x 4〜7 x 7 x 7(cm) ※新生児~2歳前後
- [M] 約8 x 8 x 8〜12 x 12 x 12(cm) ※2歳前後~8歳前後
- [L] 約13 x 13 x 13〜17 x 17 x 17(cm) ※8歳前後~
ご注文の流れ

step 1
お問い合わせ

step 2
ヒアリング
step 2
ヒアリング
メールもしくはお電話にて、製作するモデルの詳細をお聞かせいただきます。

step 3
データのご支給
step 3
データのご支給
MSCTデータ(DICOM形式)をご支給ください。ご提出いただくデータの目安として、
撮影スライス厚は1.0mm以下、画像再構成スライス間隔は0.5mm以下としてください。
※ご支給いただくCTデータは、患者様氏名・年齢を匿名化してください。
step 4
お見積書の提出
step 4
お見積書の提出
2と3をもとに、お見積りを作成し、ご提出します。
必要数量についてもお知らせください。2個目以降は安価に製作することが可能です(半年以内のご注文に限り)
step 5
ご発注
step 5
ご発注
お見積書のご承認後、注文書をご送付ください。

step 6
データの作成
step 6
データの作成
モデルの製作に必要な3Dデータを作成しご提出します。

step 7
モデルの製作
step 7
モデルの製作
3Dデータのご承認後、モデルの製作に取りかかります。

step 8
納品
step 8
納品
製品の品質を確認したうえで、ご指定の場所にモデルを納品します。
CARDIO Simuratorについてご相談のお客様は、以下の匿名化医療画像情報(個人情報)の取り扱いについてご確認頂き、
お電話、メール、お問い合わせフォームのいずれかにて、お問い合わせください。
匿名化医療画像情報(個人情報)の取り扱いについて
株式会社クロスメディカル(以下「当社」という)は、術前支援モデル「CARDIO Simulator」の製作を目的に医療機関から提供された匿名化医療画像情報を、個人情報の重要性を認識した上で保護に関する法律およびその他の諸規範を遵守し、適正な管理と保護に努めるために以下の内容についてお約束します。
- 「CARDIO Simulator」製作のために医療機関から取得した匿名化医療画像情報は、個人情報の保護に関する法令等およびその他の規範を遵守し運用します。
- 匿名化医療画像情報の運用では、個人情報保護のために適切な安全管理を実施し、紛失、き損、改ざん、漏洩等の防止に努め、不正アクセス等の防止に努めます。
- 匿名化医療画像情報は、独立した専用のPC端末およびデータ保存媒体で保管および運用し、モデル製作のための画像処理を行ないます。
- これらのPCシステムはアクセス制御と使用者認証による管理とともに、盗難防止の措置を施します。
- 匿名化医療画像情報は、「CARDIO Simulator」を製作する目的以外に利用することはありません。
- 匿名化医療画像情報は、医療機関において本人もしくは代諾者により得られる同意なしに、第三者へ提供することはありません。
- 匿名化医療画像情報は、「CARDIO Simulator」の製作終了後、次のいずれかの方法にて処理をします。
- 医療機関から送付されたCDやDVDなどのメディア媒体は、医療機関が返却を希望される場合、それらをご返却するとともに、当社に残る匿名化医療画像情報は、コンピューターやハードディスク上から完全に消去します。
- 医療機関においてメディア媒体の返却は不要とご判断された場合、当社にてそれらを無効化して廃棄するとともに、当社に残る匿名化医療画像情報は、コンピューターやハードディスク上から完全に消去します。
075-612-3900
075-612-6200
営業時間 平日9:00~18:00
CARDIO Simulator
専用お問い合わせフォーム24時間メール受付